|
So just remember, we have these six elements we discussed previously um in the videos, they have this drive where we're going to take one electron from an s orbital and promoted to one of the D orbital's so that we can create either a half filled D orbital's or totally filled, completely filled the orbital's remember this driving force that causes the exceptions within these neutral elements? Hide transcripts Now, we only have one electron here within for us, and this now becomes totally filled in and therefore more stable. We take one from the S orbital before s orbital on. If you could just get one more electron, it could become the 10. So we're gonna say here in electron and s orbital electron can be promoted to create completely filled orbital's with D nine elements. Remember, there is this need this drive by your P and D sub levels or some shells to be either half filled or totally filled. But if we look at the three d nine Orbital's, what we should notice is we just need one more electron here and it will be completely filled. So if we look at the second column, let's look at copper now, Copper, If we look at the periodic table, we'd initially think it's are gone for us to three d nine. So click on the next video and let's see what happens with them. Now that we've seen this with the first column, let's see what happens with the second column. We're not going to stay as D four when it's neutral. So again, remember, chromium has this type of exception, and the driving force is trying to get a half filled set of d orbital's. This would be the correct electron configuration of chromium. So what's gonna happen here is we're gonna take one electron from the forests and donated over here, so our three d four is going to become a D five and our for us to just gave up on electrons. But if I could somehow get one more electron in there, those deorbit will be half filled. We have only four electrons within these d orbital's. So what we're saying here is if you're doing the electron configuration of chromium, you're gonna end with a D four. Now we're gonna say an s orbital electron can be promoted to create half filled orbital's with D four electrons. But remember earlier we said that s N d sub shells or sub levels have this urge to try to be half filled or totally filled. What do we have here? We have three D with four electrons within it. Initially, we would see that it would look like are gone for us to three d four. 
So let's look at chromium if we're to determine its electron configuration. So remember exceptions start with chromium. So click on the next video and let's see what happens Hide transcripts Now that we know that these are the 60 that we have to deal with, let's see how these exceptions arise. So keep this in mind when we're looking at their electron configurations. So just remember, these are the six major types of elements where we were going to see exceptions to the electron configuration. So skip, manganese, skip iron, skip cobalt, skip nickel, and then you land on copper, where the next group of exceptions can exist. We know that's where it starts, and we're gonna skip the next four columns, right? So we start out with chromium and you skip next four. 
We're going to say that the exceptions happen with these two elements and with these four elements, so two and four we're going to say here we're gonna start out with chromium. 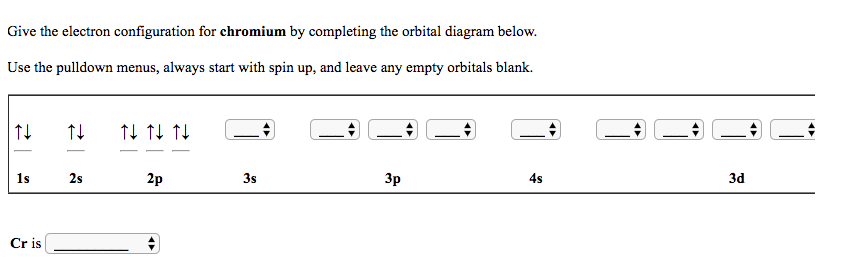
A memory tool here we can have is that chromium has an atomic number off 24. Now, when looking at exceptions to the electron configurations, we're going to say starting from chromium, which is CR as the atomic number Z increases exceptions to electron configurations can be observed. So just remember, it's this drive to either be half filled or totally filled by the P and D sub shells or sub levels that causes some of these exceptions to the electron configurations that will see later on Hide transcripts So up, up, up and come back around down, down, down, up, up, up, up, up, down, down, down, down and down. So when P sub levels or sub shells air filled halfway there, stable when d are half filled there also stable now when they're totally filled in there are also stable. So remember following off while Prince not off principle but the following the hunt's role electrons that are degenerate or have the same energy there half filled. So here, for P and G, when they're half filled, their stable in this way. We're going to say here that P and D sub shells or sub level orbital's are most stable when there are half filled or totally filled with electrons because of symmetry. Before we can talk about the exceptions to the electron configurations, it's important to understand why they exist.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
